Flowflex COVID-19 Antigen Home Test (1 Test/Kit)
$9.99 Original price was: $9.99.$4.95Current price is: $4.95.
Price per unit: $9.99 $4.95

Product Description
The Flowflex COVID-19 Antigen Home Test is a lateral flow chromatographic immunoassay intended for the qualitative detection of the nucleocapsid protein antigen from SARS-CoV-2 in anterior nasal swab specimens directly from individuals within 7 days of symptom onset or without symptoms or other epidemiological reasons to suspect COVID-19 infection.
FDA Update: Flowflex has received 510(k) clearance from the FDA, making it the first OTC rapid antigen COVID-19 test to obtain such approval. The 510(k) version of the test will also be produced domestically.
CALL FOR BULK PRICING
866-205-9215
Flowflex Covid-19 Antigen Home Test is a rapid test for the detection of SARS-CoV-2 antigens in anterior nasal specimens directly from individuals within 7 days of symptom onset or without symptoms or other epidemiological reasons to suspect COVID-19 infection.
Qualitative detection of the nucleocapsid protein antigen from SARS-CoV-2 in anterior nasal swab specimens directly from individuals within 7 days of symptom onset or without symptoms or other epidemiological reasons to suspect COVID-19 infection. This test is authorized for non-prescription home use with self-collected anterior nasal swab specimens directly from individuals aged 14 years and older or with adult-collected anterior nasal samples directly from individuals aged 2 years or older. The Flowflex COVID-19 Antigen Home Test does not require serial testing.
The Flowflex COVID-19 Antigen Home Test was compared to an FDA authorized molecular SARS-CoV-2 test. This test correctly identified 93% of positive specimens and 100% of negative specimens.
FDA Extended 21-24 Month Shelf-Life Update:
View list here: https://www.fda.gov/media/158005/download
Omicron Update:
An independent evaluation conducted by the National Institutes of Health’s (NIH) RADx program has indicated that the Flowflex™ COVID-19 Antigen Home Test does detect the Omicron subvariant BA.1 and BA.2 in live clinical samples.
Features and Benefits
- Easy and Affordable
- Highly Accurate Nasal Swab Test
- Quick Results in 15 minutes
- Safe for children as young as 2 years old
- For use with and without symptoms
- PPA (Sensitivity): 93% (95%CI: 78% – 99%)*
- NPA (Specificity): 100% (95%CI: 95% – 100%)*
*Source: FDA.gov
How Does the Flowflex COVID-19 Antigen Home Test Work?
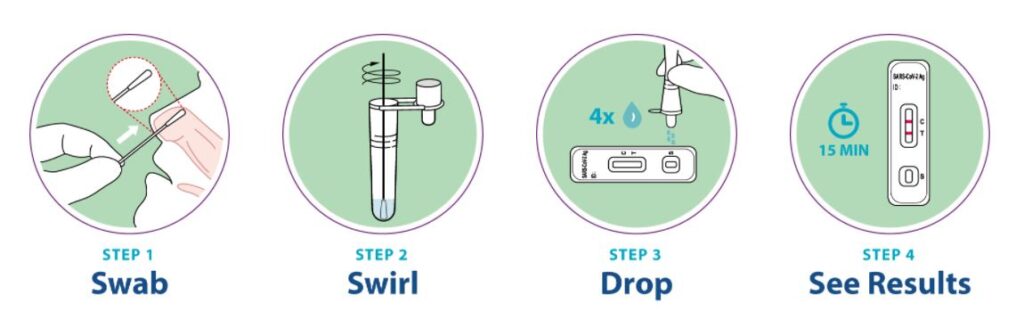
Results are for the identification of SARS-CoV-2 nucleocapsid protein antigen. This antigen is generally detectable in anterior nasal swabs during the acute phase of infection. Positive results indicate the presence of viral antigens, but clinical correlation with patient history and other diagnostic information is necessary to determine infection status. Positive results do not rule out bacterial infection or co-infection with other viruses. The agent detected may not be the definite cause of disease. Individuals who test positive should self-isolate and consult their doctor as additional testing may be necessary and for public health reporting.
• This product has not been FDA cleared or approved but has been authorized by FDA under an EUA.
• This product has been authorized only for the detection of proteins from SARS-CoV-2, not for any other viruses or pathogens.
• The emergency use of this product is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of IVDs for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Federal Food, Drug and Cosmetic Act, 21 U.S.C. § 360bbb-3(b)(1), unless the declaration is terminated, or authorization is revoked sooner.
More information about COVID-19 is available at the CDC-Centers for Disease Control and Prevention.
ref: L031-118B5
SKU: DTK-751500
Additional information
| Weight | 0.125 lbs |
|---|---|
| Dimensions | 5 × 3 × 1 in |